India's HPV Vaccination Drive: Cervical Cancer - UPSC Notes
Mar, 2026
•7 min read
Why in the News?

India has launched a free nationwide HPV vaccination drive for 1.15 crore girls aged 14 years to prevent Cervical Cancer. The campaign uses a voluntary, free-of-cost, single-dose Gardasil-4 vaccine and marks India joining 160+ countries implementing HPV vaccination programmes.
Human Papillomavirus (HPV) and Cervical Cancer
Human Papillomavirus (HPV) refers to a large group of more than 200 related viruses belonging to the Papillomaviridae family. These are non-enveloped, double-stranded DNA viruses that primarily infect epithelial cells of the skin and mucous membranes.
- Human Papillomavirus (HPV) causes Cervical cancer — the only cancer preventable by a vaccine, if administered timely.
- Most HPV infections are asymptomatic and temporary, with nearly 90% of cases clearing naturally within 1–2 years due to the body’s immune response.
- The International Agency for Research on Cancer found that 13 HPV types can cause cervical cancer in women. It affects the cervix, which connects the vagina to the upper part of the uterus.
- About 10% of women with HPV infection on their cervix will develop long-lasting HPV infections that put them at risk of cervical cancer.
Transmission
- HPV spreads mainly through intimate skin-to-skin contact, most commonly during sexual activity (vaginal, anal, or oral).
- It is considered the most common sexually transmitted infection globally.
Disease Burden in India
- Cervical cancer is the second most common cancer among women in India. Each year, the country reports around 1,20,000 new cases and 80,000 deaths annually as per GLOBOCAN 2022 data.
- India accounts for 25% of global cervical cancer deaths.
- One in every five women globally who suffer from cervical cancer is from India.
- India has the fourth-highest burden of cervical cancer morbidity worldwide.
- Persistent HPV infection accounts for nearly 85% of cervical cancer cases, with HPV-16 and HPV-18 responsible for over 80% of cases in India.
Classification of HPV Types
Human Papillomavirus types are broadly classified into low-risk and high-risk categories based on their potential to cause cancer.
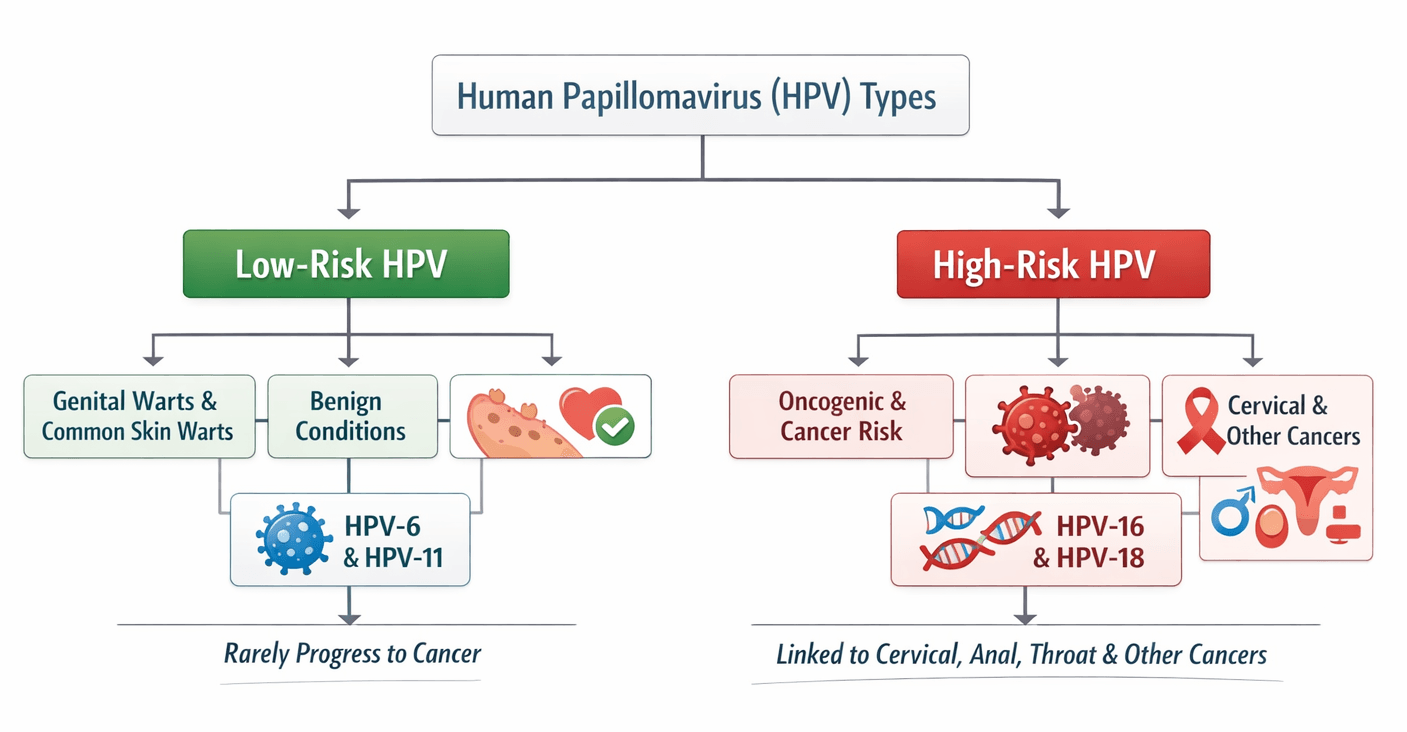
1. Low-risk HPV types:
- These strains usually cause non-cancerous (benign) conditions affecting the skin or the genital area.
- They are commonly associated with genital warts and common skin warts and rarely progress to cancer.
- The most frequently reported low-risk types are HPV-6 and HPV-11, which account for the majority of genital wart cases.
2. High-risk HPV types:
- These strains are oncogenic, meaning they can cause cancer when the infection persists for a long time.
- High-risk HPV types infect the cells of the cervix and other mucosal tissues, potentially leading to cellular changes that develop into cancer over time.
- Among them, HPV-16 and HPV-18 are the most dangerous and are responsible for the majority of HPV-related cancers, especially Cervical Cancer.
- They may also contribute to cancers of the anus, throat, vulva, vagina, and penis.
About India’s HPV Vaccination Drive
India has launched a nationwide Human Papillomavirus (HPV) vaccination programme to prevent Cervical Cancer. The initiative aims to protect adolescent girls and significantly reduce future cervical cancer cases through early vaccination.
The programme was officially launched on 28 February 2026 in Ajmer, Rajasthan, by our honourable Prime Minister Narendra Modi. Following the national launch, States and UTs across India organised their own vaccination launch events, marking the start of a national public health campaign.
- The programme initially uses Gardasil‑4, developed by Merck & Co.
- The HPV vaccination drive primarily targets around 1.15 crore girls aged 14 years across all States and Union Territories.
- The vaccine is provided free of cost at government health facilities.
- Girls who turn 15 within 90 days of the launch are also eligible under the special intensive campaign.
- The government is conducting a 90-day nationwide vaccination drive, operating daily to ensure maximum coverage.
- After the campaign period, the vaccine will continue to be administered on routine immunisation days.
With this initiative, India joins more than 160 countries worldwide that have introduced HPV vaccines in their national immunisation programmes, contributing to the global effort led by the World Health Organisation to eliminate cervical cancer as a public health problem.
Must cover this key topic: AdFalciVax: ICMR Indigenous Vaccine for Malaria
UPSC Prelims Practice Question on Cervical Cancer
QUESTION 1
Easy
Science & Technology
Which of the following viruses is primarily responsible for causing most cases of Cervical Cancer worldwide?
Select an option to attempt
The HPV Vaccine: Gardasil-4
India’s HPV vaccination programme uses Gardasil-4, a quadrivalent vaccine that protects against four major types of Human Papillomavirus (HPV). These include HPV types 16 and 18, which cause the majority of Cervical Cancer cases, and types 6 and 11, responsible for most genital warts.
The vaccine has been approved by the Central Drugs Standard Control Organisation (CDSCO) and meets strict quality, safety, and cold-chain standards required for national immunisation programmes. Under the current drive, it is administered to girls aged 14 years, ideally before the onset of sexual activity, ensuring maximum protection.
| Feature | Details |
|---|---|
| Vaccine Type | Quadrivalent HPV vaccine |
| Protects Against | HPV-16, HPV-18 (cervical cancer) and HPV-6, HPV-11 (genital warts) |
| Target Group | Girls aged 14 years |
| Dose | Single dose under the national campaign |
| Effectiveness | Around 93–100% protection against cervical cancer-causing HPV strains |
| Approval Authority | Central Drugs Standard Control Organisation (CDSCO) |
Procurement and Global Partnership
The vaccine is procured through a transparent supply mechanism in partnership with GAVI, the Vaccine Alliance. In 2023, the Government of India and GAVI established a three-year strategic partnership to strengthen India’s immunisation programme.
As part of this partnership, GAVI committed about US$250 million to:
- Identify and vaccinate “zero-dose” children (those who have not received any routine vaccines).
- Strengthen health systems and immunisation infrastructure.
- Support the introduction of new vaccines such as the HPV vaccine and the Typhoid Conjugate Vaccine (TCV).
About GAVI, the Vaccine AllianceGAVI, the Vaccine Alliance is a global public-private partnership that works to expand access to vaccines worldwide. It brings together:
The alliance focuses on protecting children from preventable diseases by improving global vaccine access and immunisation coverage. |
Must read about: Tuberculosis in India: Types, Symptoms, Challenges & Elimination Strategy 2025
HPV Vaccine Awareness: Sites & Safety Guidelines
To ensure safe and efficient delivery of the HPV vaccine, the government has established clear protocols for vaccination sites, registration, and safety under the national campaign to prevent Cervical Cancer.
- HPV vaccination is available only at designated Government Health Facilities with a functional Cold Chain Point (CCP) to maintain vaccine quality.
- Each site has a dedicated medical officer for Adverse Events Following Immunisation (AEFI) management and proper internet connectivity for digital monitoring.
- Session timings are usually 9:00 AM to 2:00 PM, including weekends and public holidays during the campaign period.
- All vaccination centres are linked to the nearest 24×7 government health facility for emergency medical support.
Safety precautions
- Girls should not be vaccinated on an empty stomach.
- Vaccination will be a single dose (0.5 ml) Intramuscular (IM) injection in the left upper arm.
- Beneficiaries are kept under 30 minutes of observation after vaccination to monitor any adverse reaction.
Registration, Recording & Monitoring
- Beneficiaries can self-register on the U-WIN digital platform, get registered through a health worker, or opt for on-site walk-in registration.
- Vaccine stock management and logistics are monitored through the Electronic Vaccine Intelligence Network (eVIN).
- Digital vaccination certificates can be downloaded from the U-WIN portal, including the nearest AEFI facility address and helpline number.
- During the three-month vaccination campaign, the left index finger of the recipient is marked after vaccination to avoid duplication.
Also see: Amoebic Meningoencephalitis: Brain-Eating Amoeba [UPSC]
Who Should Not Receive the Vaccine?
The HPV vaccine may be temporarily or permanently deferred in certain cases:
❌ Girls with moderate or severe illness (vaccination after full recovery).
❌ Individuals with a known allergy to yeast or a previous severe reaction to any vaccine.
❌ Pregnant girls or women.
❌ Girls outside the target age group
These measures ensure that the HPV vaccination programme in India remains safe, accessible, and well-monitored, supporting the broader goal of reducing cervical cancer cases.
World Cervical Cancer Elimination Day17 November is observed globally as World Cervical Cancer Elimination Day, an initiative led by the World Health Organization to raise awareness and accelerate efforts to eliminate Cervical Cancer as a public health problem. To achieve this goal, WHO has set the “90–70–90 targets” to be reached by 2030:
|
Also read: National Cancer Awareness Day 2025
How Can India Ensure the Success of the HPV Vaccination Drive?
For the nationwide HPV vaccination initiative to effectively reduce the burden of Cervical Cancer, sustained efforts in awareness, accessibility, and healthcare delivery are essential. Key measures for success are:
- Increase Public Awareness: Conduct nationwide campaigns to educate families about the link between Human Papillomavirus infection and cervical cancer, and the benefits of early vaccination.
- School-Based Vaccination Programs: Use schools as primary vaccination sites to reach adolescent girls efficiently.
- Strengthen Healthcare Infrastructure: Ensure reliable cold-chain systems, trained healthcare workers, and adequate vaccine supply at government facilities.
- Address Vaccine Hesitancy: Engage community leaders, teachers, and healthcare professionals to build trust and counter misinformation about HPV vaccines.
- Promote Screening Alongside Vaccination: Encourage regular cervical cancer screening for women to complement vaccination efforts.
- Digital Monitoring: Use platforms such as U-WIN for tracking beneficiaries, monitoring coverage, and improving programme implementation.
These steps can help India achieve higher vaccination coverage and move closer to eliminating cervical cancer as a public health problem.
UPSC Mains Practice Question on Cervical Cancer
Discuss the significance of India’s HPV vaccination programme in preventing Cervical Cancer and improving public health outcomes in the country. (150 words | 10 marks)
Evaluate your Answer in 60 SecondsConclusion
India’s nationwide HPV vaccination drive marks a major step toward preventing Cervical Cancer by promoting early immunisation, public health awareness, and women’s healthcare access.
Strengthening vaccination coverage and awareness can significantly reduce future disease burden, making this initiative highly relevant for UPSC Mains GS Paper II (Health and Social Sector Development).
Also read: International Women's Day 2026 | 8 March
Start your UPSC Preparation 2027 with SuperKalam
SuperKalam is your personal mentor for UPSC preparation, guiding you at every step of the exam journey. Practice, revise, and evaluate– all in one place.
Install the App Now


